Ultrasonic preparation of graphene
Since the special properties of graphite are known, several methods of graphite preparation have been developed. In addition to the chemical production of graphene from graphene oxide in a multi-step process, very strong oxidizing and reducing agents are required. Additionally, graphene prepared under these harsh chemical conditions often contains a large number of defects even after reduction, compared to graphene obtained from other methods. However, ultrasound is a proven alternative that can produce large quantities of high-quality graphene. The researchers developed slightly different methods using ultrasound, but in general, graphene production can be accomplished in a single step.

To give an example of a specific graphene production process: Graphite is added to a mixture of dilute organic acids, alcohols and water, and the mixture is then exposed to ultrasonic radiation. The acid acts as a "molecular wedge" separating the graphene sheets from the parent graphite. Through this simple process, a large amount of undispersed, high-quality graphene dispersed in water was produced.
Direct exfoliation of graphene
Ultrasound can prepare graphene in organic solvents, surfactant/aqueous solutions or ionic liquids. This means that strong oxidizing or reducing agents can be avoided. (2007) produced graphene by exfoliation under the action of ultrasound. The melt of graphene oxide at a concentration of 1 mg/ml was sonicated. AFM images showed that there were always flakes with uniform thickness (1 nm), and there was no graphene thicker than 1 nm or less than 1 nm thick in these good graphene oxide exfoliated samples flakes, it was concluded that under these conditions, complete exfoliation of graphene oxide to obtain individual graphene oxide flakes was achieved.
Preparation of graphene sheets
A large number of pure graphene sheets were successfully prepared during the production of non-stoichiometric TiO2 graphene nanocomposites by thermally hydrolyzing the suspension with graphene nanosheets and titania peroxide complexes. Pure graphene nanosheets are made of natural graphite using a high-intensity cavitation field generated by an ultrasonic processor in a high-pressure ultrasonic reactor at 5 bar. The resulting graphene sheets have high specific surface area and unique electronic properties, which can be used as a good support for TiO2 to enhance the photocatalytic activity. The quality of the ultrasonically prepared graphene is much higher than that obtained by the Hummer method, in which the graphite is exfoliated and oxidized. Since the physical conditions inside the ultrasonic reactor can be precisely controlled, and by assuming that the concentration of graphene as a dopant will vary in the range of 1–0.001%, it is possible to produce graphene in a commercial-scale continuous system.

Ultrasonic treatment of graphene oxide
Fabrication process for graphene oxide (GO) layers using ultrasonic radiation. Twenty-five mg of graphene oxide powder was suspended in 200 mL of deionized water. A homogeneous brown suspension was obtained by stirring. The resulting suspension was sonicated (30 min, 1.3 × 105 J), and after drying (373 K), sonicated graphene oxide was prepared. FTIR spectra showed that sonication did not change the functional groups of graphene oxide.
Functionalization of graphene sheets
Xu and Suslick (2011) described a one-step method for the preparation of polystyrene-functionalized graphite. In their study, they used graphite flakes and styrene as basic raw materials. By sonicating graphite flakes in styrene (a reactive monomer), ultrasonic radiation causes mechanochemical exfoliation of graphite flakes into monolayer and few-layer graphene flakes. At the same time, functionalization of graphene flakes with polystyrene chains was also achieved. The same functionalization process can also be performed with other graphene-based composite vinyl monomers.
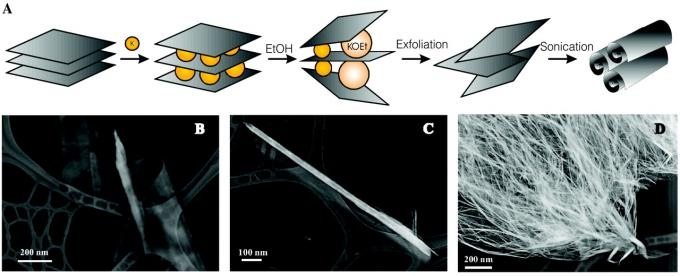
Preparation of carbon nanorolls
Carbon nanocoils are similar to multi-walled carbon nanotubes. The difference with multi-walled carbon nanotubes is the open tip and the complete accessibility of the inner surface to other molecules. Wet chemical synthesis by intercalation of graphite with potassium, exfoliation in water, and sonication of the colloidal suspension. Ultrasound-assisted rolling up of graphene monolayers into carbon nanotubes (Fig. 3) yields a conversion efficiency as high as 80%, making the production of nanotubes a hot spot for commercial applications.
Graphene Dispersion
The dispersion grade of graphene and graphene oxide is extremely important to exploit the full potential of graphene and its specific properties. If graphene is not dispersed under controlled conditions, the polydispersity of graphene dispersions, once incorporated into the device, can lead to unpredictable or non-ideal effects because the properties of graphene vary with its structural parameters. Sonication is a proven treatment method that attenuates interlaminar forces and allows precise control of important processing parameters. "For graphene oxide (GO), which is typically exfoliated into monolayer sheets, one of the main polydispersity challenges is due to the variation in the lateral area of the flakes. By varying the graphite feedstock and sonication conditions, the average lateral dimension of GO can vary from 400 nm is shifted to 20 μm. Ultrasonic dispersion of graphene has been demonstrated in many other studies to produce fine or even colloidal slurries.
Zhang et al (2010) have shown that by using ultrasonic waves, stable graphene dispersions with a high concentration of 1 mg·mL-1 and relatively pure graphene sheets can be obtained, and the as-prepared graphene sheets have 712 S·m1 high conductivity. Fourier transform infrared spectroscopy and Raman spectroscopy test results show that the ultrasonic preparation method has less damage to the chemical and crystal structure of graphene










